Our Competitive Advantage
Unlock the power of innovation through a unique business model that integrates dedicated business units and specialized technical support units — positioning COS at the forefront of clinical outcomes research with an unmatched strategic advantage.

Scientific
Business Units
Our dedicated business units bring deep expertise, scientific rigor, and innovative thinking to help you stay ahead in healthcare research.
-
The Qualitative Science business unit is your partner in developing, validating, and implementing COAs that truly matter.
With a deep understanding of patient-reported outcomes, we guide you through the intricate process of capturing meaningful data that reflects the patient experience.
Our expertise ensures that your trials and interventions have reliable and sensitive measures, fostering confident decision-making and regulatory success.
-
The Quantitative Science business unit stands at the forefront of data analysis and interpretation.
Our team of statistical experts harnesses data to uncover meaningful insights, transforming complex information into actionable recommendations.
With a commitment to precision and innovation, we ensure that your clinical outcomes research benefits from rigorous quantitative analysis, enabling informed decisions and robust outcomes.
-
The Prospective Studies business unit — also known as Mixed-Methods — is dedicated to uncovering new perspectives in clinical outcomes research.
We design and execute prospective studies that provide real-time insights into the effectiveness and safety of interventions.
With a focus on robust methodologies, our team navigates the complexities of prospective research, delivering data that empowers evidence-based decision-making and helps shape the future of healthcare.
-
The Evidence Synthesis business unit brings together qualitative insights, quantitative data, and regulatory intelligence to create the most compelling, evidence-based narratives for product approval and labeling success.
We sit at the intersection of science, strategy, and storytelling — helping clients make the strongest possible case for their clinical outcomes.
-
The Research Operations provides the operational backbone that keeps complex COA research running smoothly, on time, and to the highest quality standards.
Our team leads project management, study coordination, and data management—bringing structure, consistency, and oversight across every phase of a study. From startup through closeout, we manage timelines, vendors, documentation, and day-to-day execution so scientific teams can stay focused on delivering strong evidence and sponsor-ready results.
Key benefits of cross- functional
scientific business units.
-
Each business unit provides custom-tailored services and solutions to meet the unique needs of each sponsor, ensuring the highest—quality outcomes.
-
Our researchers provide extensive experience and industry knowledge — offering innovative solutions and strategies that help navigate complex challenges and drive successful outcomes.
-
With our data-driven approach, we are committed to delivering the highest quality results that enable our sponsors to have the information they need in today’s complex healthcare landscape.
-
We are committed to providing sponsors with the most reliable and comprehensive evidence and reports — ensuring that our sponsors have what they need to make informed decisions.
-
Our director-led approach offers a distinct advantage by leveraging the expertise and leadership of our experienced directors. We ensure that our research initiatives are guided by a deep understanding of the industry, cutting-edge methodologies, and strategic insights.
-
Each business unit is backed by TSUs, departments that supply a broad range of skills and expertise to support the unit’s related needs and streamline operations.
-
Our services, solutions, and strategic partnerships offer clients and sponsors complete coverage across the COA research landscape — eliminating the need to engage with outside vendors and saving you time and money.
Clinical Outcomes Solutions
Organizational Structure

-
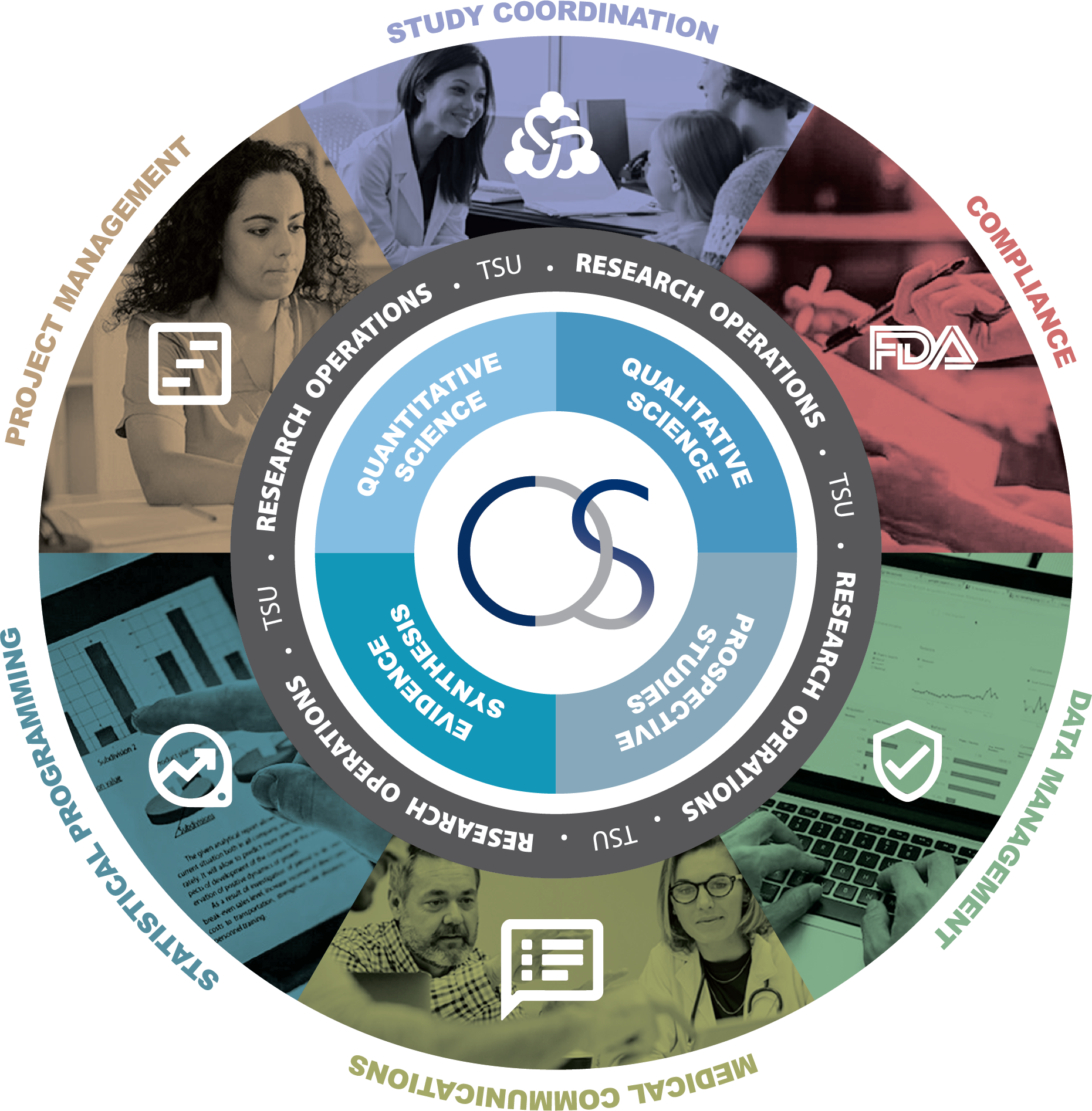
The above diagram presents Clinical Outcomes Solutions’ model as a sponsor-centered, integrated ecosystem built around four interconnected business units — Quantitative Science, Qualitative Science, Prospective Studies, and Evidence Synthesis — shown as overlapping rings to emphasize cross-functional collaboration.
Within each business unit, work is led by Director-Led Research Teams (represented by the people icons), ensuring scientific leadership, accountability, and strategic alignment. Surrounding and enabling these teams are the Technical Support Units (TSUs) (represented by the green icons), which provide the operational and regulatory backbone required to deliver efficiently and at high quality—Study Coordination, Project Management, Statistical Programming, Medical Communications, Data Management, and FDA Compliance.
The intersections between the rings represent deliberate collaboration across business units, while the central overlap highlights integrated delivery where multiple disciplines come together. At the core, the Sponsor benefits from this coordinated structure—combining specialized scientific leadership with scalable TSU support to accelerate evidence generation, maintain compliance, and deliver cohesive, end-to-end clinical outcomes solutions.

Backed by World-Class
Technical Support Units.
Our Technical Support Units play a pivotal role in augmenting and reinforcing the capabilities of each of our Business Units — empowering each business unit to oversee studies comprehensively from inception to completion.
-
Our Study Coordination team provides the structure, oversight, and operational discipline needed to keep complex research programs moving forward with confidence. From study planning and resource allocation to timeline management, risk mitigation, budget oversight, and stakeholder communication, we ensure every phase of the study is aligned, efficient, and execution-ready. By maintaining rigorous quality standards, managing change proactively, and delivering clear documentation and reporting throughout the study lifecycle, we help sponsors stay on track, reduce operational burden, and achieve successful study delivery.
-
Our Project Management team provides the strategic oversight, structure, and accountability needed to successfully execute complex research initiatives. From defining scope, timelines, resources, and budgets to proactively managing risk, quality, change, and stakeholder communication, we keep every aspect of the project aligned and moving forward. Through disciplined planning, ongoing monitoring, and clear reporting, we help sponsors reduce operational complexity, maintain momentum, and deliver studies on time, on budget, and to the highest standards.
-
Our Statistical Programming team transforms complex clinical data into high-quality, analysis-ready outputs that support confident decision-making and reliable study reporting. From developing and validating SAS programs to integrating data across sources, generating tables, listings, and figures, and applying rigorous quality control standards, we ensure accuracy, consistency, and efficiency throughout the reporting process. With deep expertise in regulatory expectations, CDISC standards, and data visualization, we help sponsors deliver clear, compliant, and submission-ready results.
-
Our Medical Communications team translates complex scientific data into clear, credible, and impactful communications that support both strategic objectives and scientific integrity. From manuscripts, abstracts, posters, and scientific reports to conference materials, publication planning, and scientific training, we develop high-quality content tailored to diverse audiences. By combining rigorous scientific review, clear messaging, and a strong understanding of regulatory requirements, we help sponsors communicate study findings with accuracy, confidence, and influence.
-
Our Data Management team ensures study data is accurate, secure, and fit for purpose from collection through archiving. From database design and data capture to cleaning, verification, quality control, and reporting, we build the systems and processes needed to maintain data integrity at every stage. With a strong focus on regulatory compliance, privacy, and operational efficiency, we help sponsors generate reliable, high-quality data that supports confident decision-making and successful study execution.
-
Our Compliance team helps ensure that every aspect of our work is grounded in quality, integrity, and accountability. From regulatory and data privacy compliance to quality standards, ethical practices, and marketing governance, we establish the frameworks, safeguards, and oversight needed to support consistent, compliant operations. Through clear policies, rigorous standards, and ongoing education and training, we help protect stakeholder trust, reduce risk, and reinforce confidence in the quality of our services and deliverables.
Key benefits of
Technical Support Units.
-
TSUs allow for streamlined and efficient study management — reducing the time and cost required to complete each study and increasing overall productivity.
-
TSUs ensure premium quality research and reports with rigorous quality assurance, meticulous attention to detail, and advanced data analysis — delivering exceptional outcomes and evidence-based recommendations.
-
TSUs offer specialized expertise, leveraging a team of experienced professionals with in-depth knowledge and skills in their respective fields — ensuring comprehensive and accurate support for diverse research needs.
-
TSUs mitigate the risks associated with study coordination, project management, data management, and SAS programming — minimizing the potential for errors or delays that could impact study outcomes.
-
TSUs ensure consistency and standardization, maintaining high-quality standards and protocols across studies for reliable, comparable outcomes supporting accurate analysis and decision-making.
-
TSUs provide flexible and adaptable solutions, enabling us to respond to changes swiftly, integrate new methodologies, and customize services to optimize research outcomes for each study’s unique needs.
-
TSUs enhance communication by providing concise documentation, facilitating collaboration, ensuring regulatory compliance, and offering project management support.
Clinical Outcomes Solutions
Clinical Research Process

-
The updated diagram presents Clinical Outcomes Solutions’ model as a sponsor-centered, integrated ecosystem built around four interconnected business units—Quantitative Science, Qualitative Science, Prospective Studies, and Evidence Synthesis—shown as overlapping rings to emphasize cross-functional collaboration.
Within each business unit, work is led by Director-Led Research Teams (represented by the people icons), ensuring scientific leadership, accountability, and strategic alignment. Surrounding and enabling these teams are the Technical Support Units (TSUs) (represented by the green icons), which provide the operational and regulatory backbone required to deliver efficiently and at high quality—Study Coordination, Project Management, Statistical Programming, Medical Communications, Data Management, and FDA Compliance.
The intersections between the rings represent deliberate collaboration across business units, while the central overlap highlights integrated delivery where multiple disciplines come together. At the core, the Sponsor benefits from this coordinated structure—combining specialized scientific leadership with scalable TSU support to accelerate evidence generation, maintain compliance, and deliver cohesive, end-to-end clinical outcomes solutions.
